Retatrutide is the most discussed research compound in UK weight loss and metabolic research circles right now — but many people searching for it are still unclear on exactly what it is, how it works and where it stands in the regulatory pipeline. This complete guide answers all of those questions.
Retatrutide Definition
Retatrutide (research code: LY3437943) is a synthetic peptide developed by Eli Lilly and Company. It is classified as a triple receptor agonist — meaning it simultaneously activates three different hormone receptors in the body: GLP-1 (Glucagon-like peptide-1), GIP (Glucose-dependent insulinotropic polypeptide) and Glucagon receptors.
This triple mechanism is what makes retatrutide unique and distinguishes it from every other compound in its class. No other approved obesity compound activates all three receptors simultaneously.
How Does Retatrutide Work?
- GLP-1 receptor: Reduces appetite, slows gastric emptying, improves insulin secretion. The mechanism shared with semaglutide (Ozempic/Wegovy).
- GIP receptor: Enhances insulin response, supports fat metabolism, may reduce GLP-1-related nausea. The second mechanism added by tirzepatide (Mounjaro).
- Glucagon receptor: Increases resting energy expenditure — the body burns more calories at rest. This is the entirely new mechanism that only retatrutide possesses.
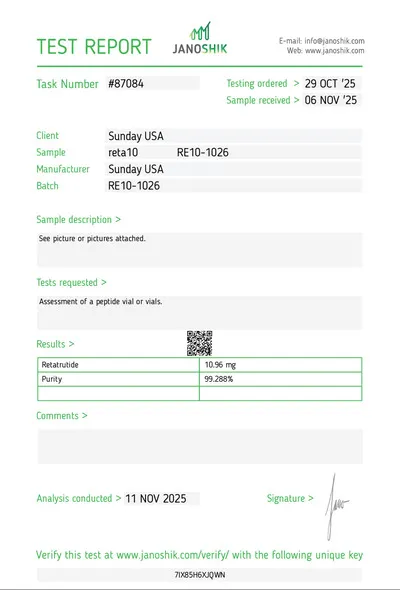
Retatrutide Clinical Data
The landmark Phase 2 trial, published in the New England Journal of Medicine in June 2023, showed: at the 12mg dose, participants lost an average of 24.2% of body weight at 48 weeks. Critically — weight loss had not plateaued when the trial ended at 48 weeks, suggesting even greater reductions with longer treatment duration.
UK Regulatory Status 2026
As of April 2026, retatrutide is in Phase 3 TRIUMPH trials. MHRA submission is expected early 2027. MHRA approval optimistically 2027–2028. NHS availability: 2029 at the earliest. Research-grade retatrutide is available now from verified UK suppliers for R&D purposes.
Order from Jeo Peptides Supply
Janoshik verified. UK cold chain delivery. Personal WhatsApp response. R&D use only.