The UK's two most researched GLP-1 class compounds are now both available for research: retatrutide (Synedica) and tirzepatide (the active ingredient in Mounjaro). Understanding the difference is essential for researchers choosing between them.
Mechanism Comparison
| Compound | Brand | Receptors | Mechanism |
|---|---|---|---|
| Semaglutide | Ozempic / Wegovy | GLP-1 only | Single agonist |
| Tirzepatide | Mounjaro / Zepbound | GLP-1 + GIP | Dual agonist |
| Retatrutide | Synedica | GLP-1 + GIP + Glucagon | Triple agonist |
Clinical Efficacy Data
- Semaglutide (STEP trials): ~15% average weight reduction at 68 weeks. GLP-1 only.
- Tirzepatide/Mounjaro (SURMOUNT): ~20.9% at 72 weeks. GLP-1 + GIP dual action.
- Retatrutide (NEJM Phase 2): 24.2% at 48 weeks — AND weight loss plateau not yet reached. GLP-1 + GIP + Glucagon.
Side Effect Profiles
All three compounds share a similar GI side effect profile — nausea, diarrhoea, constipation — common to GLP-1 agonists. Tirzepatide's GIP component partially offsets GLP-1-related nausea. Retatrutide's additional glucagon component may increase nausea at higher doses in some research subjects.
UK Research Availability 2026
Mounjaro (tirzepatide) received NICE TA1026 approval and NHS rollout began June 2025 for eligible patients. Retatrutide remains in Phase 3 TRIUMPH trials — MHRA approval expected 2027 at the earliest. Both are available from Jeo Peptides Supply as research-grade compounds for R&D use only.
Order from Jeo Peptides Supply
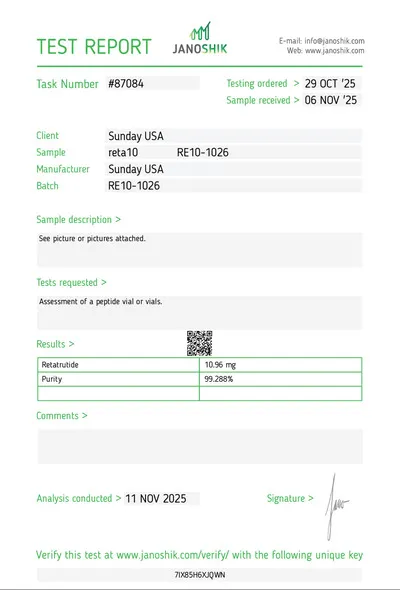
Janoshik verified. UK cold chain delivery. Personal WhatsApp response. R&D use only.